 Nintedanib falls under the small molecule tyrosine-kinase inhibitor, targeting vascular endothelial growth factor receptor, fibroblast growth factor receptor and platelet derived growth factor receptor. On 15'th October 2014, US Food and Drug Administration granted the approval to this small molecule tyrosine-kinase inhibitor in order to treat idiopathic pulmonary fibrosis, and got a positive perspective from the EMA on 20'th November 2014, being approved in the European Union in the month of January, 2015. In June 2011 until 15 October 2021, the US Food and Drug Administration granted the orphan drug designation to Nintedanib in order to treat idiopathic pulmonary fibrosis. In the year of 2014, in European Union, this medication (nintedanib) was approved as a combination therapy in order to treat NSCLC, and is approved for this indication in certain other regions of the world. In July 2016 until 6 September 2019, the US FDA granted orphan drug designation to nintedanib in order to treat systemic sclerosis (including the associated interstitial lung disease). Nintedanib was granted priority review designation by the Food and Drug Administration prior to being approved in the United States on 6'th September 2019, in order to slow the rate of decline in pulmonary function in patients with SSc-ILD (systemic sclerosis associated interstitial lung disease). It is specifically the first FDA-approved treatment for this rare lung condition. The US FDA granted priority review designation to nintedanib as well as breakthrough therapy designation. The Food and Drug Administration granted the approval of Ofev (Nintedanib) to Boehringer Ingelheim Pharmaceuticals, Inc. The nintedanib 150 mg price is around 25,985 INR for a supply of 10 capsules, depending on the pharmacy/wholesaler you visit. Any WHO-GDP & ISO certified pharmaceutical wholesaler company would be best to buy nintedanib.
1 Comment
The benefit of adding daratumumab to the PI-based triplet bortezomib, lenalidomide, and dexamethasone for newly diagnosed patients with multiple myeloma who underwent high dose melphalan chemo and autologous hemopoietic cell transplant was assessed.
In order to assess the safety and effectiveness of carfilzomib, lenalidomide, dexamethasone, daratumumab combination therapy for newly diagnosed patients with multiple myeloma, in the absence of high-dose melphalan chemo and autologous hemopoietic cell transplant. According to the clinical and correlative pilot carried out study at the Memorial Sloan Kettering Cancer Center in New York, New York, Newly diagnosed Patients with multiple myeloma were enrolled b/w October 1, 2018, and November 15, 2019. The median follow up from the initiation of the treatment was noted as 20.3 months (95 percent CI, 19.2 to 21.9 months). Eight 28-day cycles along with the intravenous carfilzomib, 20/56 mg/m2 (days 1, 8, and 15); oral lenalidomide, 25 mg, (days 1 to 21); dexamethasone, 40 mg weekly, orally or intravenously (cycles 1 to 4), and 20 mg following cycle 4; and intravenous daratumumab, 16 mg/kg (days 1, 8, 15, and 22 [cycles 1 to 2]; days 1 and 15 [cycles 3 to 6]; and day 1 [cycles 7 and 8]). The observed primary endpoint was the MRD (minimal residual disease) rate, in absence of high-dose melphalan chemo and autologous hemopoietic cell transplant. Secondary endpoints included determining safety as well as tolerability, evaluation rates of the clinical response per the International Myeloma Working Group, and estimating PFS and the overall survival rates. Total of 41 assessable patients were enrolled (median age, 59 years; range, 30 to 70 years); 25 (61 percent) enrolled patients were female, and 20 (49 percent) had high-risk MM. Primary endpoint (minimal residual disease negativity in the bone marrow; 10 to 5 sensitivity) was reached in 29 of 41 patients (71%; 95% CI, 54 percent to 83 percent), and thus the trial was deemed completed. The median time to minimal residual disease negativity was six cycles (range, 1 to 8 cycles). Secondary end points of the overall response rate and the VGPR or CR rate were 100 percent (41 of 41 patients) and 95 percent (39 of 41 patients), respectively. At eleven months of the median follow-up, the one year progression free survival rate and the overall survival rate was noted as 98 percent (95 percent CI, 93 percent to 100 percent) and 100 percent, respectively. The most commonly reported grade 3/4 adverse events were neutropenia, rash, lung infection, and increased alanine aminotransferase level. Though, no casualties were reported. In this nonrandomized clinical trial, carfilzomib, lenalidomide, dexamethasone, daratumumab combination therapy was linked with the high rates of minimal residual disease negativity in newly diagnosed patients with the multiple myeloma and the high rates of progression-free survival. Reference: https://pubmed.ncbi.nlm.nih.gov/33856405/ Medication for the treatment for the types of psoriasis and psoriatic arthritis is known by the name Apremilast, mostly sold under the name of otezla. Certain types of psoriasis plague are commonly caused due to the downregulation of immune activity within the skin. The changes in the skin result in epidermal thickening, redness, and itchiness, and scaly plaques.
Development History: The CADTH Canadian Drug Expert Committee (CDEC), in July 2015 recommended that apremilast not be listed for the treatment of adult patients with moderate-to-severe. The main reasons for the recommendation included the lack of comparative evidence with other treatments and inadequate evidence to evaluate the use of apremilast in the patient. Approval history: 21 March 2014, the US Food and Drug Administration (FDA) approved apremilast to treat Psoriatic Arthritis, moderate to severe plaque psoriasis, oral ulcers. It is an oral inhibitor of PDE4 (phosphodiesterase 4). FDA approval was based on safety, double-blind Phase study evaluates apremilast on 207 adult patients with active oral ulcers. The early-stage or first recommendation was at the April meeting, and again was a request for reconsideration that was considered at the July 2015 meeting. Dosage and usage: Approximately 7 million people in the United States get effects with arthritic inflammation, psoriatic arthritis. Psoriatic, a chronic disease, acts on skin with thick patches, scaly skin results in excessive growth of skin cells. As, now apremilast work on different skin and on different cells the dosage is also prescribes in apremilast 10mg/apremilast 20 mg/apremilast 30mg. Mostly, the combination of different users is also advised by the doctor as:
Now the question arises how to take apremilast tablets? It is advisable to take it according to the doctor's prescription. They can be taken with or without food- morning and evening orally through the mouth. It is recommended to take tablets without crushing, splitting, or chewing the tablets. Price: Apremilast comes in different trade brands. The apremilast price of the brand name Aprezo is approximately Rs. 295 (10 tablets). Note:- This is a prescription medicine, while buying online and offline keep a doctor prescription copy handy.
Tofacitinib specifically is an oral, small molecule JAK inhibitor being investigated for Ulcerative Colitis.
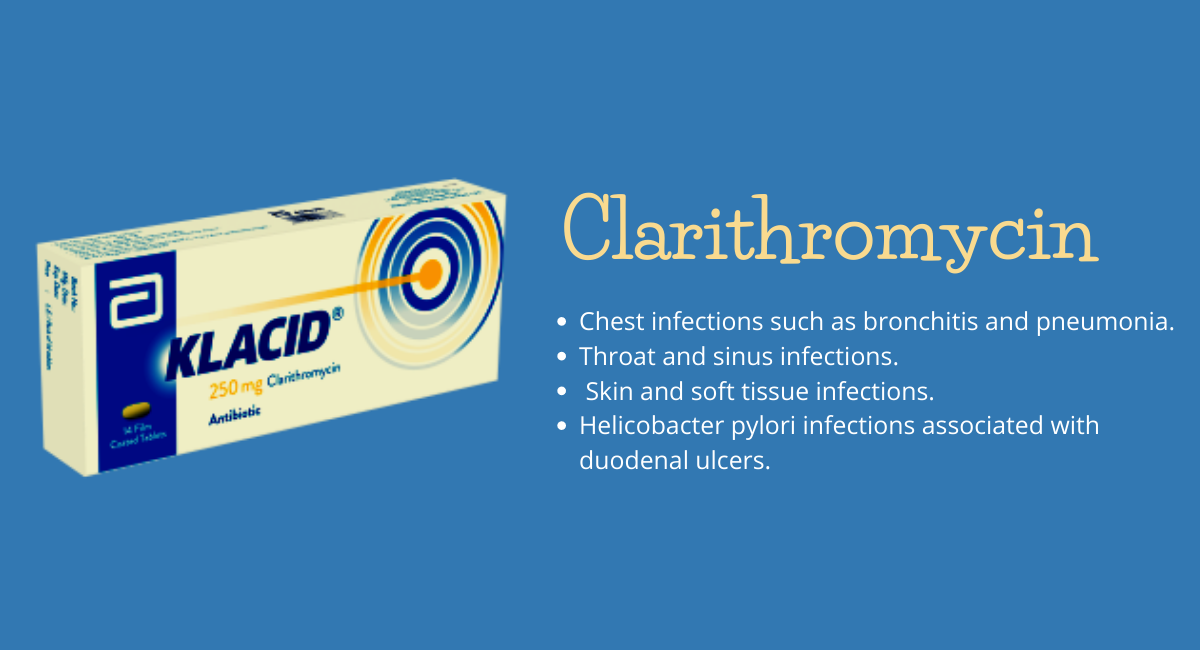
Findings from the post-hoc analysis suggests that the treatment with tofacitinib (Xeljanz) demonstrated to be rapidly effective for the moderate/severe ulcerative colitis (UC), even in those patients who previously failed TNF inhibition. According to William Sandborn, MD, of the University of California San Diego, "The oral small-molecule JAK inhibitor had higher efficacy than placebo for induction as well as maintenance of remission, rates of herpes zoster were numerically higher in the tofacitinib recipients, regardless of the previous response to the tumor necrosis factor inhibition. Tumor Necrosis Factor inhibitor failure has been specified as a predictor of the weak prognosis in the ulcerative colitis. Patients who are unmanageable to this therapy may be challenging to treat, and for that reason have an unmet need for the alternative therapies. Regardless of how, "Our findings demonstrate that tofacitinib (Xeljanz) induces and also manages the clinical response, endoscopic enhancement, and remission in the patients either with and without prior to the failure of the tumor necrosis factor," the team described in the CGH (Clinical Gastroenterology and Hepatology. At the beginning of this year, the Sandborn's group described the efficacy of tofacitinib (Xeljanz) vs certain biologics in ulcerative colitis. The ongoing investigation involved participants in OCTAVE Induction, Sustain, and Open trials. The clinical findings and this follow-up examination were favored by the manufacturer of tofacitinib named Pfizer. The author noted, "At 8'th week (induction) and 52'nd week (maintenance), tofacitinib demonstrated good efficacy in comparison to placebo, in any case of sort of non-response to tumor necrosis factor inhibition and number of unsuccessful therapies." While induction, the effects of treatment were noted normally identical for patients with the previous failure of tumor necrosis factor inhibitor and also those without failures, and for those patients who failed 1 and 2 or multiple tumor necrosis factor inhibitors. Moving on to maintenance, treatment effects were noted numerically greater for tofacitinib 10 mg in comparison to the tofacitinib 5 mg dose, in any case of the theindianpharma.com/buy-xeljanz-tofacitinib-online/previous tumor necrosis factor inhibition unsuccessful status. Also while maintenance, patients with earlier failure demonstrated identical treatment effects for the endoscopic improvement and remission with Xeljanz 5 mg whether they had a either primary or secondary non-response. In order to double the tofacitinib dose, the recaptured clinical response for maximum patients who lost the response with 5 mg dose by month 2, and this effect was sustained for three years. Reference: https://www.healio.com/news/gastroenterology/20210310/xeljanz-effective-for-uc-regardless-of-prior-tnfi-failure https://www.pharmastar.it/news/gastro/tofacitinib-efficace-in-pazienti-con-colite-ulcerosa-che-hanno-fallito-altri-trattamenti-34862 https://www.medpagetoday.com/gastroenterology/inflammatoryboweldisease/91612  According to a statement which was came out in a regulatory filing by Natco Pharma, "Our marketing partner named BPI (Breckenridge Pharmaceutical Inc) has got the final approval for its ANDA, which stands for Abbreviated New Drug Application for everolimus tablets (a generic version of Afinitor) from the US Food and Drug Administration (USFDA). Natco's partner Breckenridge Pharmaceutical Inc (BPI) aims to launch 7.5 mg, 5 mg, and 2.5 mg strengths of the xeverolimus tablets presently within the upcoming few weeks. According to Natco Pharma, "The launching of everolimus as the strength of 10 mg is completely subject to the confidential terms of a settlement and licence agreement entered into with the owner of the brand Afinitor." "The launch date of everolimus as the strength of 10 mg will be declared after a certain period of time,'' the company added. The above mentioned strengths of Everolimus are indicated in order to treat breast cancer and certain other types of cancers. On behalf of industry sales data, Natco Pharma stated Afinitor and its therapeutic equivalents had given rise to annual sales of about 712 million USD in the United State of America during the 12 months ending December 2020. Natco Pharma's shares were trading 6.45 % higher at Rs 886.50 apiece on the BSE. Reference: https://www.financialexpress.com/lifestyle/health/natcos-marketing-partner-gets-usfda-nod-for-cancer-treatment-drug/2208320/ What Clarithromycin is and for what it is used for?
Clarithromycin belongs to a group of medicine named macrolide antibiotics. Antibiotics are able to block the growth of bacteria which are responsible for causing infections. The clarithromycin uses are as follows:
What is clarithromycin available as?
When should I give clarithromycin? Clarithromycin usually should be given twice each day, once in the morning and once in the evening. It should be given at about the same times each particular day so that it will be helpful in order to remember. How to take Clarithromycin? This medicine always should be taken exactly as your healthcare practitioner has instructed you. Clarithromycin is consumed with or without food. The usual clarithromycin dosage is 250 mg twice a day. The usual dose is 250 mg two times daily. In case of severe infections, your healthcare provider can increase the clarithromycin dose to 500 mg two times daily. Treatment may take 7 to 14 days and last minimum a couple of days once the symptoms are gone. Possible Side Effects: Like all medicines, klacid can also cause side effects, although not every individual gets them. The commonly observed side effects may include:
Warnings and Precautions: Interact with your healthcare practitioner prior to start taking clarithromycin 500 mg tablet if:
What are some medications that need to be used with caution?
When should the medicine start working? This medication often starts working after taking the medicine for 2 or 3 days. It is crucial to take the full course of treatment that has been prescribed. Avoid stopping early. In case your child has a fever (temperature above 38°C) that does not improve, or they become more unwell, contact your healthcare practitioner. What if my child is sick (vomits)?
What if I forget to give it? If you remember up to 4 hours following you should have given a dose, give your child the forgotten dose. What if I give too much Clarithromycin? Generally, Clarithromycin is a safe agent. It is unlikely not responsible for causing any problems in case of excess dose taken by mistake. In an event of excess clarithromycin dose, contact your healthcare practitioner. How should Clarithromycin be stored? Tablets as well as oral suspension needs to be stored in a cool (15°C to 30°C) dry place, protected from the light and out of the reach of infants/children. How much does clarithromycin cost? In India, the cost of clarithromycin is quite reasonable. Prices may vary depending upon branded to non-branded drugs varying from around 100 INR to 1300 INR. NOTE: The piece of information provided in this article is just for informational purposes and is not served as a substitute for the medical treatment, consultation, diagnosis, of a qualified healthcare practitioner. Essential Thrombocythemia specifically is a chronic condition characterised by the excess platelets in the blood. It is a group of conditions named myeloproliferative neoplasms (MPNs), which also includes myelofibrosis (MF) and polycythaemia vera (PV).
Symptoms of ET: In patients with Essential Thrombocythemia generally do not have any signs when they are diagnosed. These patients are identified after abnormal outcomes while on a routine complete blood test for anything else. In case symptoms appear, they tend to do so as time passes. The common essential thrombocythemia symptoms include:
What causes Essential Thrombocythemia: The essential thrombocytosis is a typical chronic myeloproliferative disorder. In this disorder, the bone marrow produces excess cells that mainly create platelets. The thrombocythemia causes are exactly not specified. Approximately 90% of people with this complication have a gene mutation contributing to disease. In case an underlying complication such as an infection or iron deficiency causes a high platelet count, it's known as secondary thrombocytosis. In comparison to essential thrombocythemia, the secondary thrombocytosis mainly causes less risk of blood clots as well as bleeding. How is ET diagnosed: The essential Thrombocythemia is mainly diagnosed with the help of laboratory tests including: Blood tests: It is able to identify an increase in the blood cells and exclude other causes of a high cell count. Gene mutation analysis: It is used in order to detect any gene mutations you may have, specifically JAK2, CALR or MPL. Bone marrow investigations: You may be prescribed either for a bone marrow aspiration or both an aspiration and bone marrow biopsy. This is performed in order to look for the classic signs of Essential Thrombocytosis. What is the prognosis of ET? The essential thrombocytosis prognosis may vary widely. Your haematologist is the appropriate person to instruct you on behalf of your individual circumstances. Treatment: A hematologist may recommend the specific essential thrombocytosis treatment as well as management. Those patients who are with reduced risk for clotting are basically observed without applying any therapy; low-dose aspirin may be used. Those patients who are with higher risk for clotting need medical therapy in order to decrease platelets to normal levels, and are prescribed for low-dose aspirin in order to protect clotting. Drug Therapy: Hydroxyurea (Hydrea), Anagrelide (Agrylin) and Interferon alfa (immediate-release preparations Intron A and Roferon-A and sustained-release preparations PEG-Intron and Pegasys), are the most widely prescribed medications for ET. Hydroxyurea (Hydrea): As a myelosuppressive agent, this medication can be prescribed as initial therapy for Essential Thrombocytosis. Hydroxyurea 500 mg is taken orally, and often effective in order to decrease the platelet count within a few weeks, with some short-term adverse reactions. Side effects of this drug may include:
Anagrelide (Agrylin): As a non cytotoxic agent, it is effective in order to decrease the platelet formation. The anagrelide capsules should be consumed by mouth. Some side effects of anagrelide may occur, including:
Low-dose aspirin: Aspirin is recommended to be given orally. It is useful for patients who are at the high risk for the clotting problems. In those patients who are with reduced risk for clotting, proof for its uses are comparatively less strong. This drug may also be responsible in order to increase the risk of bleeding in those patients who are with excess platelets. Plateletpheresis: In this process, a kind of special machine is used in order to skim the platelets in blood and then return the plasma and red cells to the patient. It is prescribed only in the emergency circumstances, such as complications of acute clotting, in case of extremely high platelet count and require to be reduced rapidly. JAK2 inhibitors: This helps in blocking the function of the JAK2 mutation which involves in order to slow down the blood cell production, reduces the spleen size and helps improve symptoms. Approximately 55% of patients with essential thrombocytosis have been detected to have a mutation named JAK2. Busulfan: This is usually recommended to be given to patients who have adverse reactions when consuming hydroxyurea. It may be prescribed as a tablet. Like hydroxyurea, busulfan directly affects the bone marrow and may be responsible for a fall in the blood cell counts. The common adverse reactions of busulfan include:
NOTE: Prior to starting any kind of treatment, consult a healthcare provider. In order to know more about any drug, read full PI (Prescribing Information). This chemotherapy drug is manufactured & marketed by the company named Eli Lilly under the brand name Alimta. It is prescribed with cisplatin in order to treat patients with the malignant pleural mesothelioma whose disease is unrespectable or curative surgery can not be implemented. It can also be used in the treatment of non-small cell lung cancer.
This medication is recommended to be given as an infusion into the vein (intravenous, IV). Precautions:
Warnings:
Imatinib specifically is a TKI (Tyrosine Kinase Inhibitor). A kinase is basically an enzyme that promotes the growth of cells. There are many sorts of kinases, which can control the different phases of cell growth. By blocking a particular enzyme from working, Imatinib 400 mg is able to resist the cancer cell growth.
It acts by blocking signals within cancer cells, which involves in preventing a series of chemical reactions that permits the cell in order to grow and divide. Imatinib comes in the form of tablets, and you should take it by mouth. Your dose may be dependent on your diagnosis. Each dose is consumed with a glass of water and a meal. Precautions:
Warnings:
|
AuthorMy Name is Amit Sharma I am a pharmacist, I am having 25+ years of experience in Pharma medications. ArchivesCategories
All
|



 RSS Feed
RSS Feed
